46 – Preparing for a Clinical Trial
24th July 2023
Bit by bit, I’m getting there.
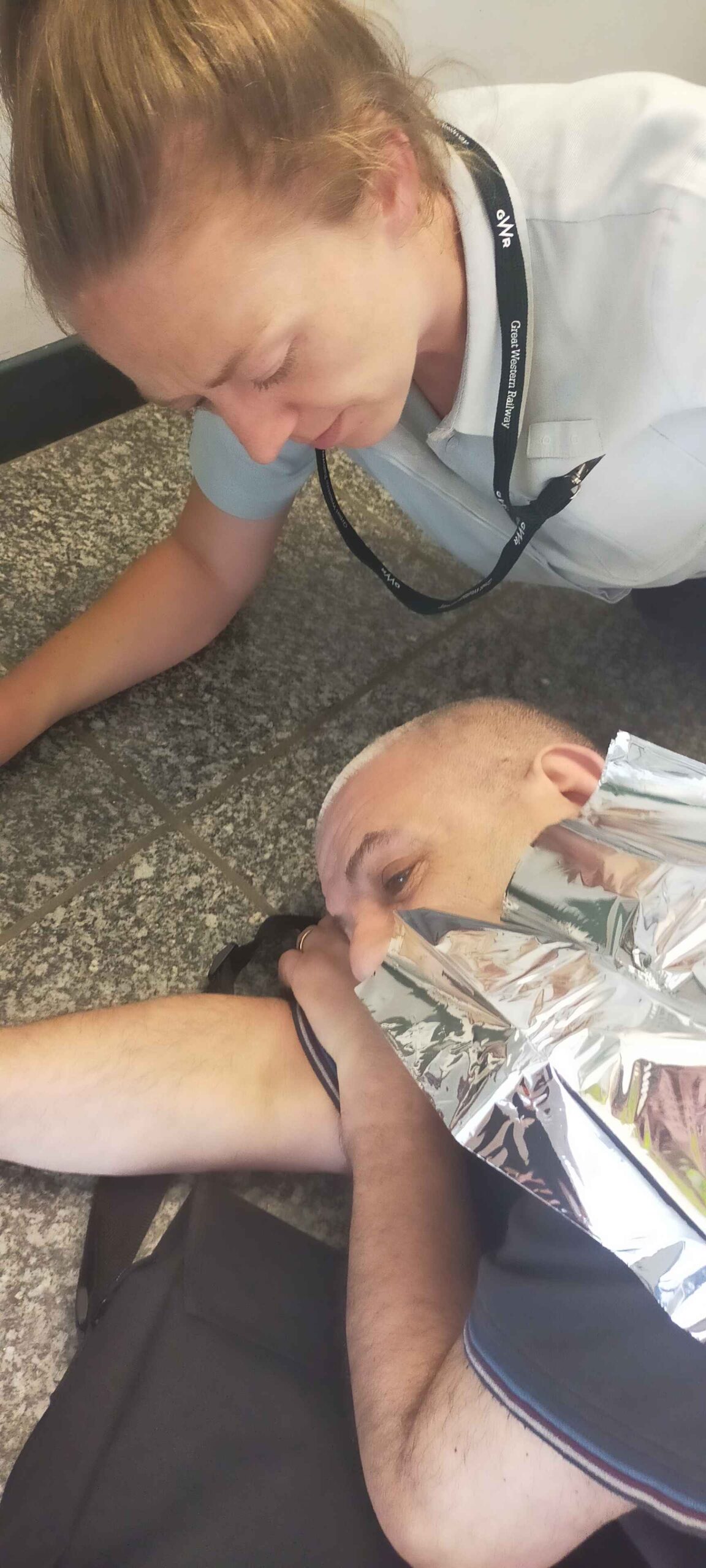
I previously mentioned the mishap at Gloucester Railway station on 22nd June, when I collapsed on my way to probably my most important meeting of this year – a pre-trial consultation to see if I qualify for the screening process and subsequent entry to a clinical trial, to test some potential new cancer treatments.
And so, a week later on 29th June, I did make it to the meeting with Professor Harrington and a couple of his research team. My eldest daughter Rachel came with me, ostensibly to take notes – but in reality, to catch me if I decided to keel over again.
Thankfully I stayed on my feet – unaided

The consultation started exactly on time at The Royal Marsden Hospital, in London. No hanging about, which can happen and just adds to stress levels.
Prof Harrington is clearly a leader in his field, but had a very easy-going manner about how he interviewed me. And we went through a lot, from my cancer history to prior lifestyle, current medications and a physical checkup, Then the meeting continued with a detailed explanation of the proposed clinical trial and allowed me to work through my list of questions, which were either answered there and then, or certainly in the 20+ page of trial description, treatment scheduling that I was given.
Everything looked good to me, but I wasn’t permitted to say ‘yes’ there and then. I had to consider the trial paperwork and then return to hospital a few days later (4th July), to initial and sign consent in person.
Very professional and correct.
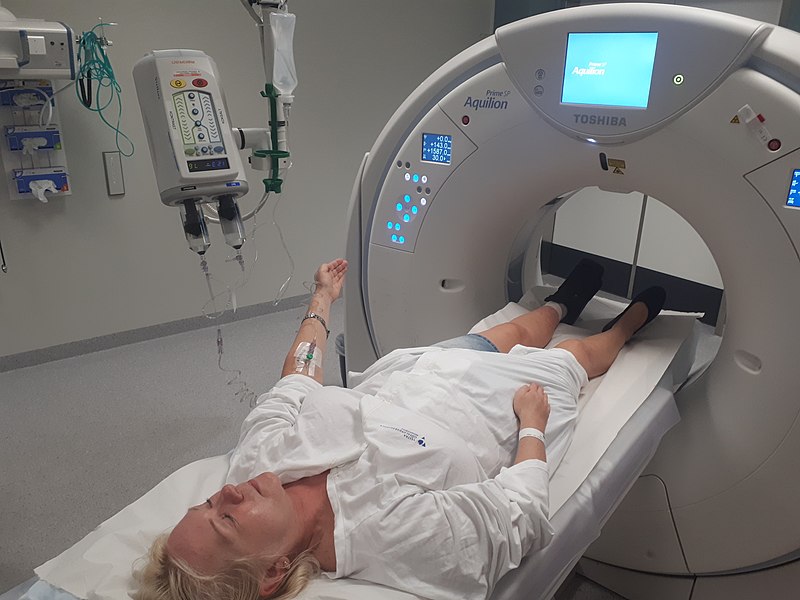
Once the consent was signed, we started again on a full physical examination, blood samples taken, ECG and finally a ‘baseline’ CT scan with contrast was undertaken. Baseline means a reference scan - so that all future scans and imaging of my tumours can be compared back to this original.
CT with contrast is actually two scans. The first pass in the scanner is without the dye, then a small pump injects the dye into a vein and a second pass in the scanner is done. The dye helps to improve the images and highlight cancerous areas within the body.
Unfortunately, when injecting the contrast dye into my veins, the dye leaked out into the tissues under my skin, this is called ‘extravasation’. It’s not serious, but pretty uncomfortable for a few hours – and then a couple days for the swelling to subside.
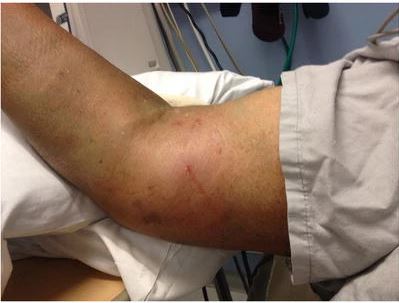
So, this meant another visit on 7th July to re-scan me. Thankfully this went well, and all of my screening interviews / test results / scans etc have been sent to the drug company that is behind this particular trial for the final decision on my suitability to enter the trial.
I received positive confirmation a week or so ago and this coming week, I begin treatments in what is honestly something with an unknown outcome, but very much worth doing in any case.
I have a full schedule, with various treatments and tests on days 1, 8, 29, 50 and 71. Following this there are subsequent treatment intervals of around 3 weeks, with comparative CT scans every nine weeks.
All in all then, while it's been a pretty hectic month for me with a few trips to London, I'm very glad to be on this trial and starting soon.
But also I'm pragmatic enough to know and understand that a clinical trial is just that - a trial.
And it's primarily in place to test the drug(s). The patients on a trial I suppose, always hope this might just be the 'miracle drug'. Realistically though. it'll simply provide data and other information for future development of cancer treatments.
But for me, right now - after having two longish lines of treatment without success and being faced with a third (and probably final) course of chemotherapy, this trial allows me an additional line, while being extremely well looked after.
And that can’t be bad at all 🙂